Tone of Voice Matters in Neural Communication! Study from MBL Neurobiology Shows How

Contact: dkenney@mbl.edu; 508-685-3525 WOODS HOLE, Mass. --
WOODS HOLE, Mass. -- The dialogue between neurons is of critical importance for all nervous system activities, from breathing to sensing, thinking to running. Yet neuronal communication is so fast, and at such small scale, that it is exceedingly difficult to explain precisely how it occurs. A preliminary observation in the Neurobiology course at the Marine Biological Laboratory (MBL), enabled by a custom imaging system, has led to a clear understanding of how neurons communicate with each other by modulating the “tone” of their signal, which previously had eluded the field. The report, led by Grant F. Kusick and Shigeki Watanabe of Johns Hopkins University School of Medicine, is published this week in Nature Neuroscience.
In 2016 Watanabe, then on the Neurobiology course faculty, introduced students to the debate over how many synaptic vesicles can fuse in response to one action potential (see this 2-minute video for a quick brush-up on neurotransmission). To probe this controversy, they used a “zap-and-freeze” imaging technology conceived by co-authors M. Wayne Davis, Watanabe and Erik Jorgensen, and built by Leica for testing in the Neurobiology course. They zapped a neuron with electricity to induce an action potential, then quickly froze the neuron and took an image. They saw multiple vesicles fusing at once at many synapses, the first novel finding of this Nature Neuroscience report.
 Shigeki Watanabe, left, and Erik Jorgensen with the flash-and-freeze imaging system, a forerunner to zap-and-freeze. Both systems were developed in the Jorgensen lab and are descendants of the 1979 “freeze slammer” invented by John Heuser, Tom Reese and colleagues. Credit: Cveta Tomova
Shigeki Watanabe, left, and Erik Jorgensen with the flash-and-freeze imaging system, a forerunner to zap-and-freeze. Both systems were developed in the Jorgensen lab and are descendants of the 1979 “freeze slammer” invented by John Heuser, Tom Reese and colleagues. Credit: Cveta TomovaBut there was more. Back at Johns Hopkins, Kusick and Watanabe decided to walk through the neurotransmission process with zap-and-freeze, taking images every 3 milliseconds after the action potential. That’s when they found an answer to an even larger question – how do neurons change the tone of their neurotransmission signal?
At any given time, only a few synaptic vesicles are in “docked” position, meaning loaded and ready to release neurotransmitter. Immediately after an action potential, the number of docked vesicles decreases by 40 percent, so after 2 to 3 action potentials, the docked vesicles would be depleted. (That is, their signal or “voice” would become weaker and weaker, as more action potentials are induced.) But they found that, within 14 milliseconds following an action potential, new vesicles are swiftly recruited to the docked pool that can fuse and release neurotransmitter, and this recruitment is transient such that neurotransmission can be strong or weak on a millisecond time scale. This is the first close-up look at neural communication that adds up from a temporal perspective.
“What this means is that we have identified a mechanism that neurons use to communicate through intonations,” Watanabe says. “Each docked vesicle is like a word that neurons can use for communication at any given moment. It has been known for decades that neurons can speak more than a few words at a time, and they can also change the tone of these words. The question was how. We’ve shown that neurons continuously bring in more words, but by simply changing the number of vesicles, they can raise or lower the voice. If you are asking a question, you will raise the intonation at the end of a sentence – neurons do so by changing the number of docked vesicles ready to go.”
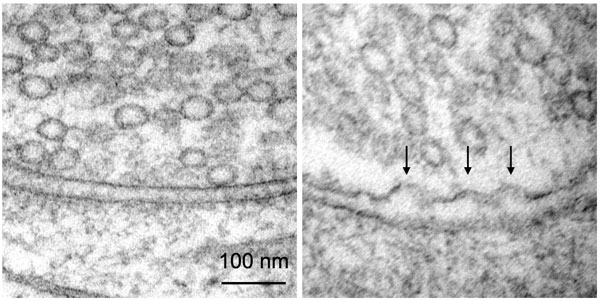 Synapses before (left) and after stimulation, with arrows showing the moment of neurons talking to each other (fusion of synaptic vesicles). Credit: MBL Neurobiology course students Kristina Lippmann, Kadidia Adula, Thien Vu, and Eddie Hujber
Synapses before (left) and after stimulation, with arrows showing the moment of neurons talking to each other (fusion of synaptic vesicles). Credit: MBL Neurobiology course students Kristina Lippmann, Kadidia Adula, Thien Vu, and Eddie Hujber The “zap and freeze” electron microscopy technology is the 21st-century version of the “freeze slammer” developed by John Heuser, Tom Reese et al., and used at MBL nearly 50 years ago to demonstrate how neurons communicate with each other.
The MBL Neurobiology course faculty and students co-authoring this paper include Jorgensen and Davis (University of Utah), Kristina Lippmann (University of Leipzg), Kandidia P. Adula (University of California, Los Angeles), and Edward J. Hujber and Thien Vu (University of Utah). Watanabe, who was a 2014 Grass Fellow at the MBL, returns nearly annually to teach in the Neurobiology course and as a Whitman Center scientist.
 Left to right: Grant Kusick (Watanabe lab), MBL Neurobiology course students Christine Prater and Stephen Lee, and Shigeki Watanabe in Woods Hole in 2019.
Left to right: Grant Kusick (Watanabe lab), MBL Neurobiology course students Christine Prater and Stephen Lee, and Shigeki Watanabe in Woods Hole in 2019.Citation:
Grant F. Kusick, M. Chin, S. Raychaudhuri, K. Lippmann, K. P. Adula, E.J. Hujber, T. Vu, M.W. Davis, E.M. Jorgensen, S. Watanabe (2020) Synaptic vesicles transiently dock to refill release sites. Nature Neuroscience, DOI: 10.1038/s41593-020-00716-1
—###—
The Marine Biological Laboratory (MBL) is dedicated to scientific discovery – exploring fundamental biology, understanding marine biodiversity and the environment, and informing the human condition through research and education. Founded in Woods Hole, Massachusetts in 1888, the MBL is a private, nonprofit institution and an affiliate of the University of Chicago.